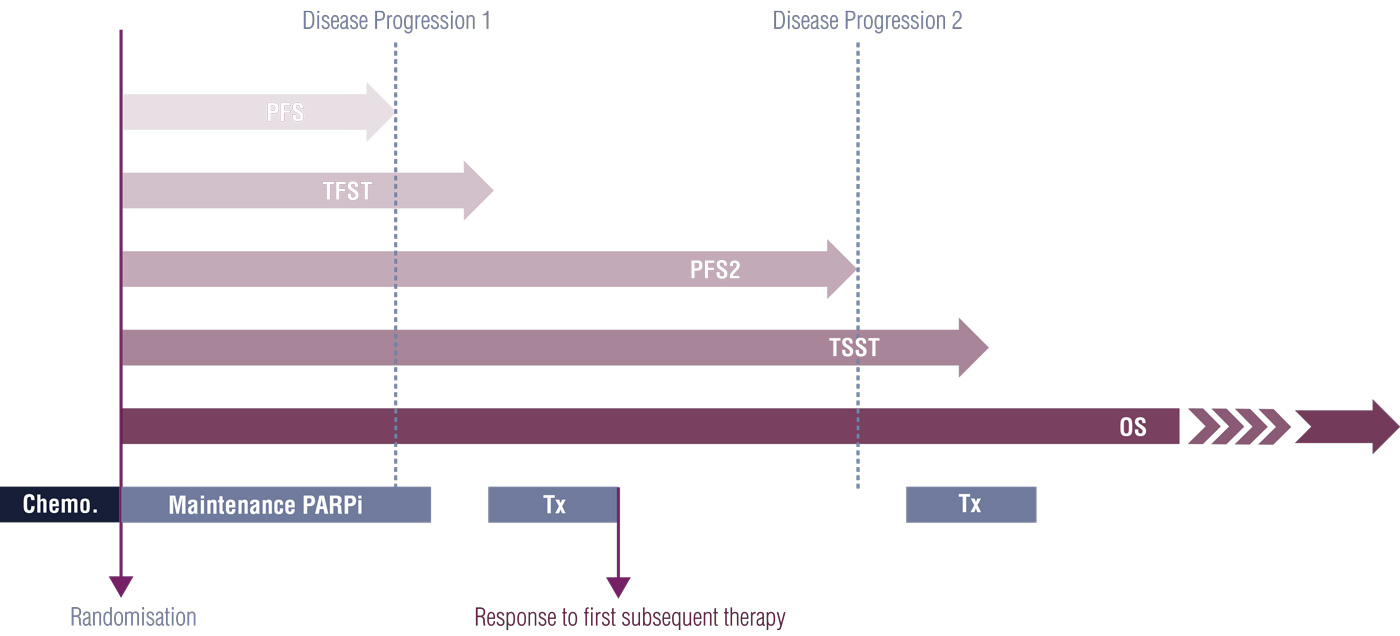
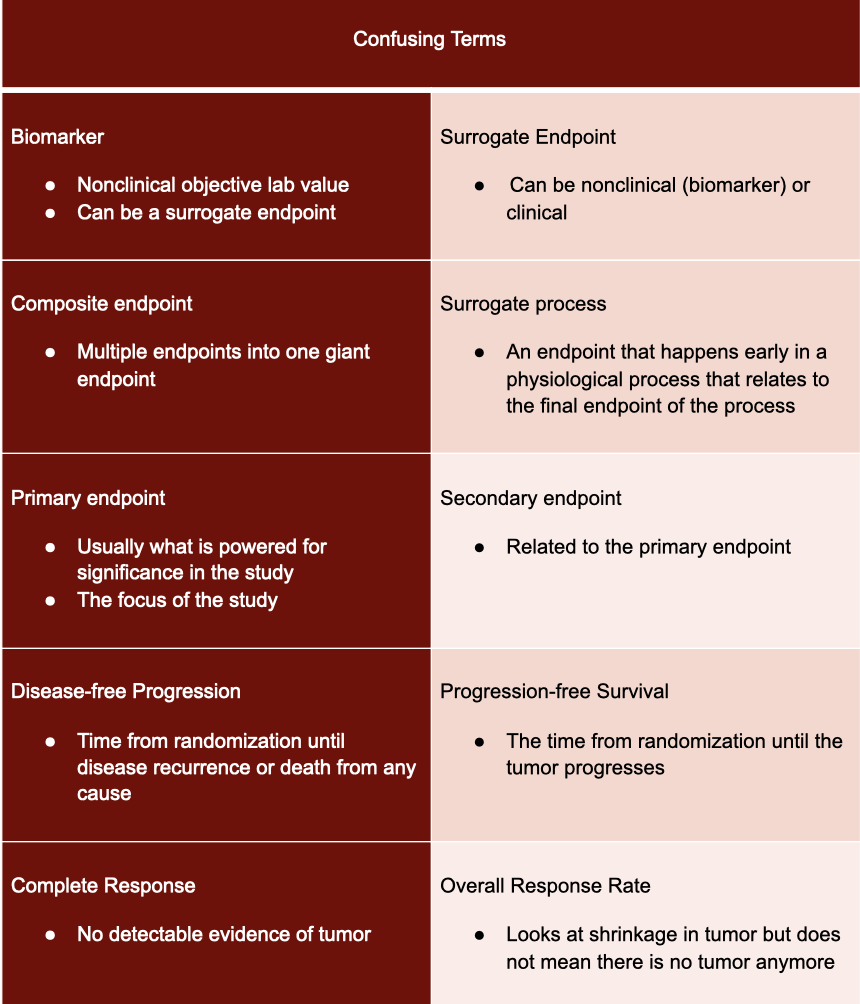
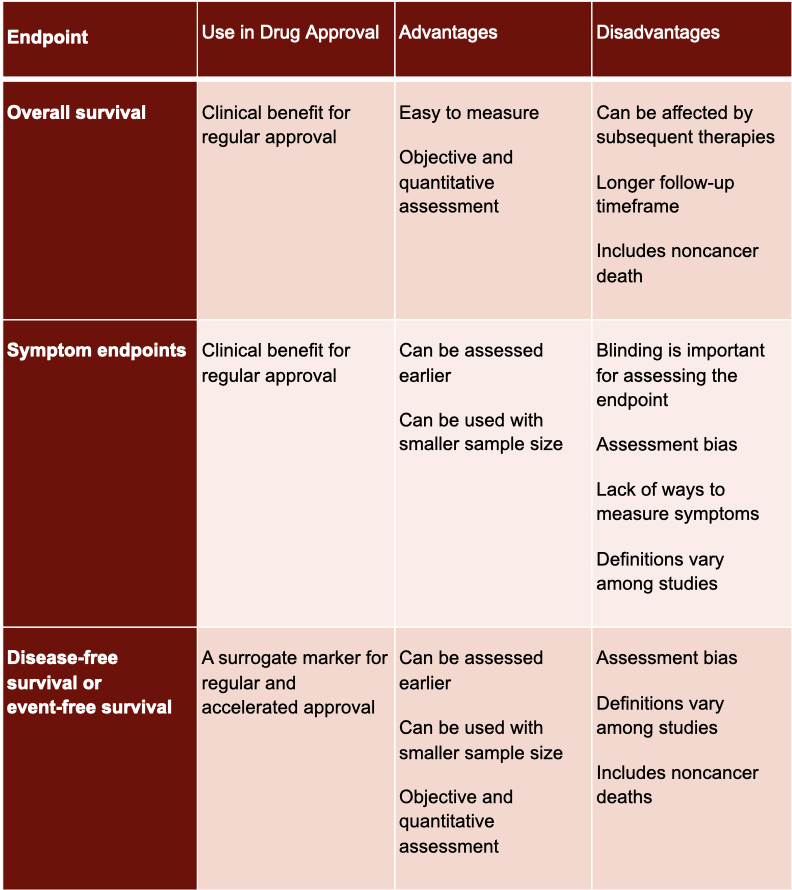
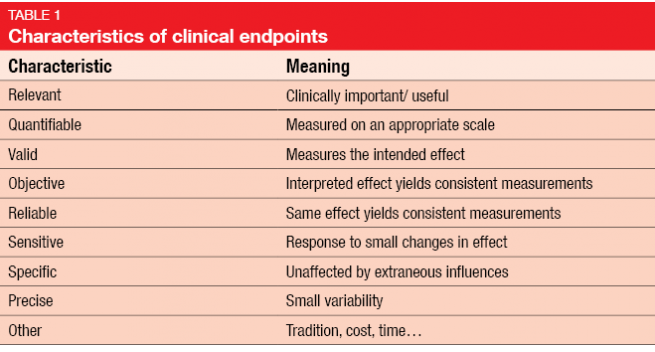
Explanation of primary endpoints in acute myeloid leukemia clinical trials. | Download Scientific Diagram
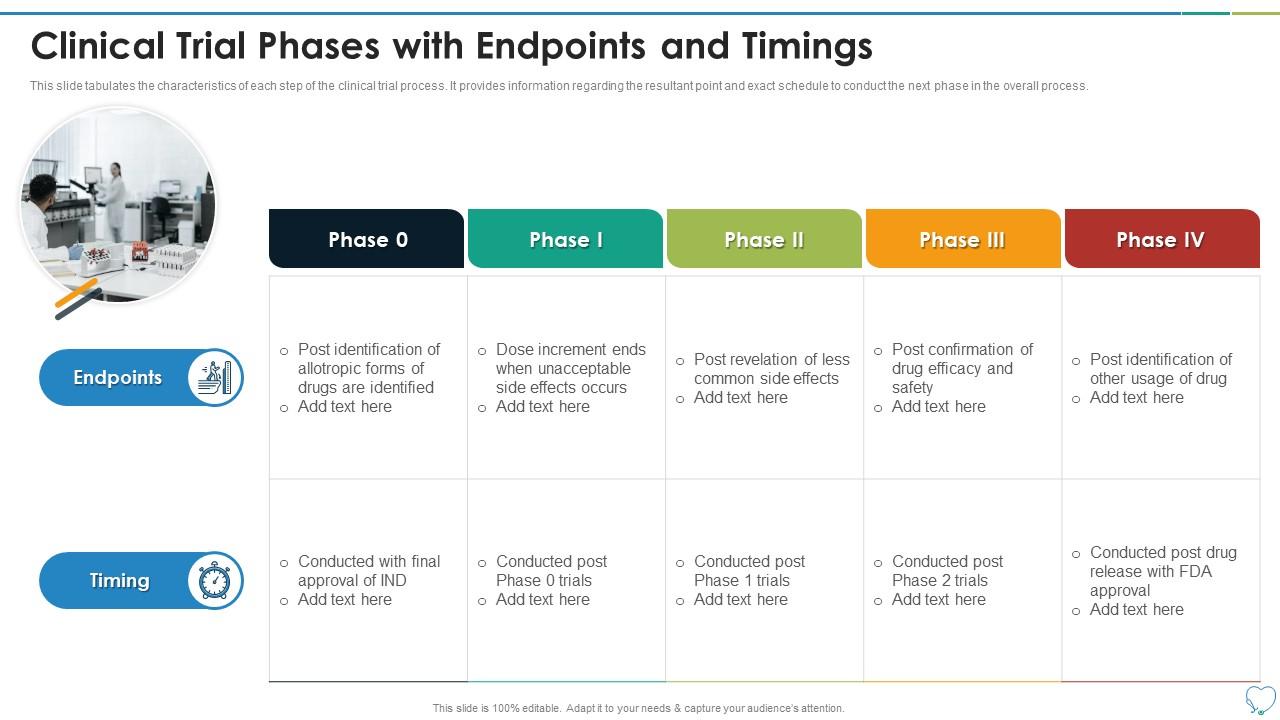
Clinical Trial Phases Endpoints And Timings | Presentation Graphics | Presentation PowerPoint Example | Slide Templates
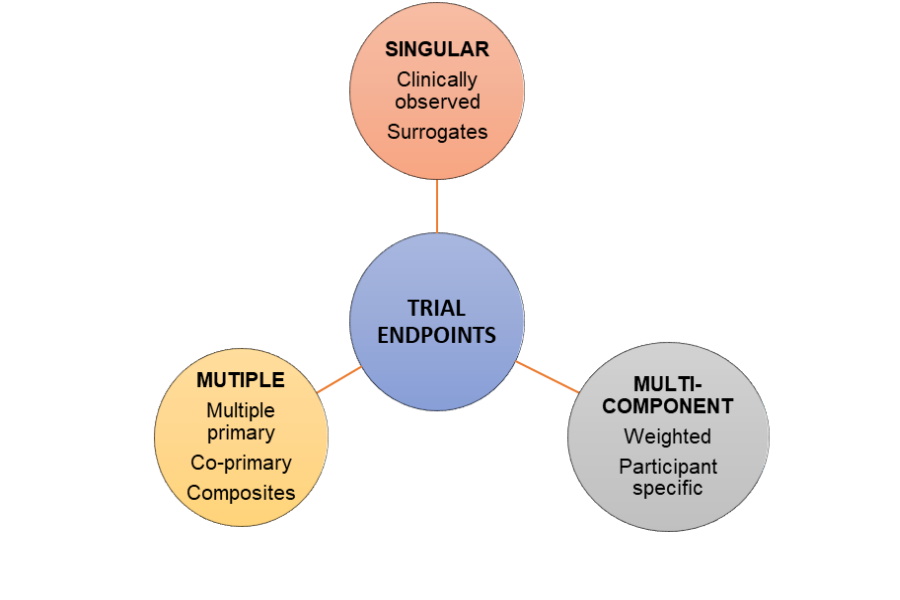
The Endpoint Selection: a Complex Process in the Clinical Trials Design Page CRA School | The International Clinical Research Academy Page | CRA School | The International Clinical Research Academy
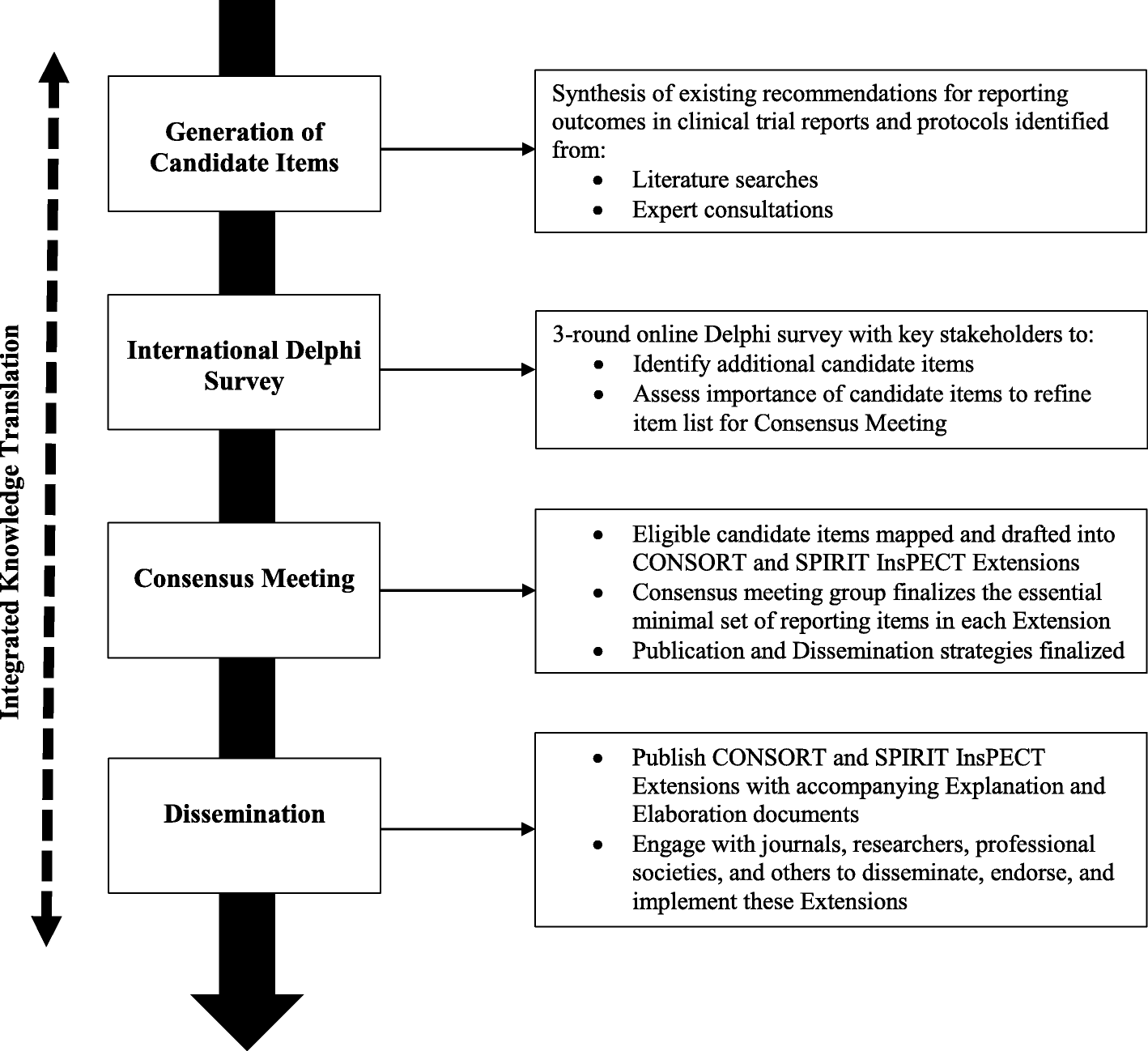
Improving outcome reporting in clinical trial reports and protocols: study protocol for the Instrument for reporting Planned Endpoints in Clinical Trials (InsPECT) | Trials | Full Text

Development of Novel, Value-Based, Digital Endpoints for Clinical Trials: A Structured Approach Toward Fit-for-Purpose Validation | Pharmacological Reviews