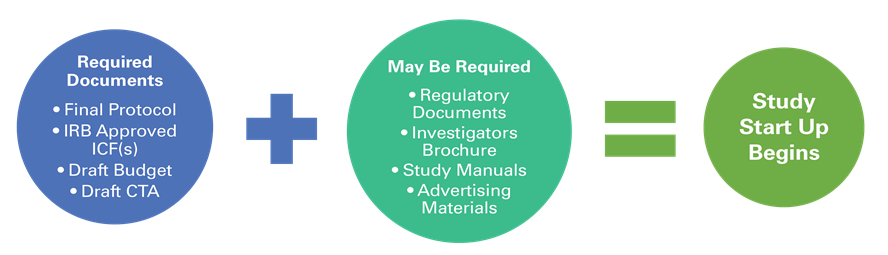
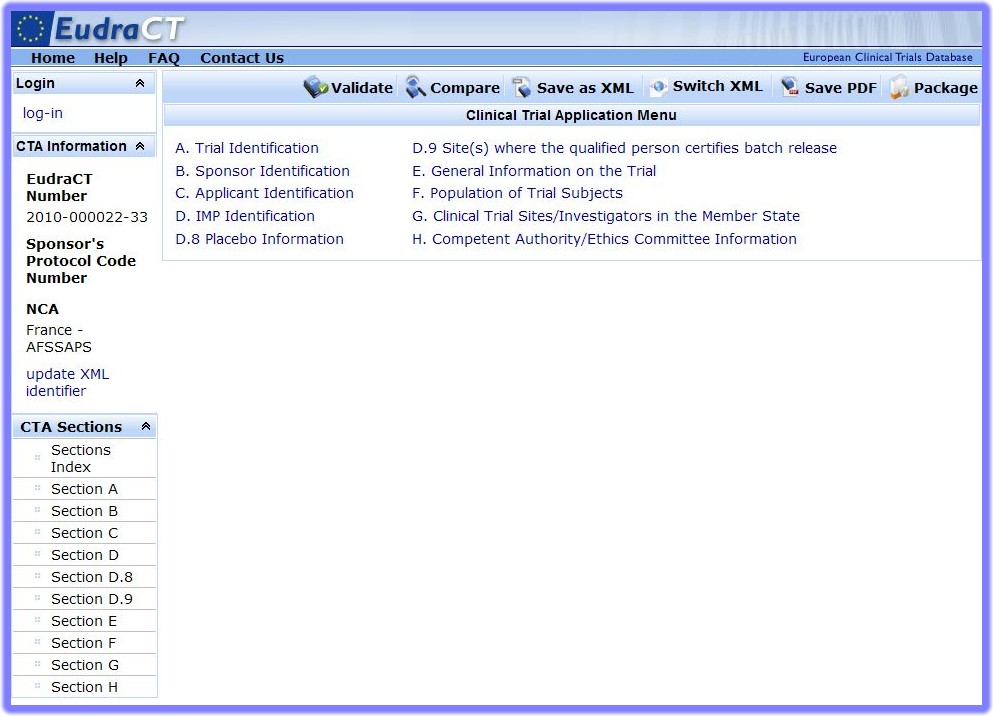
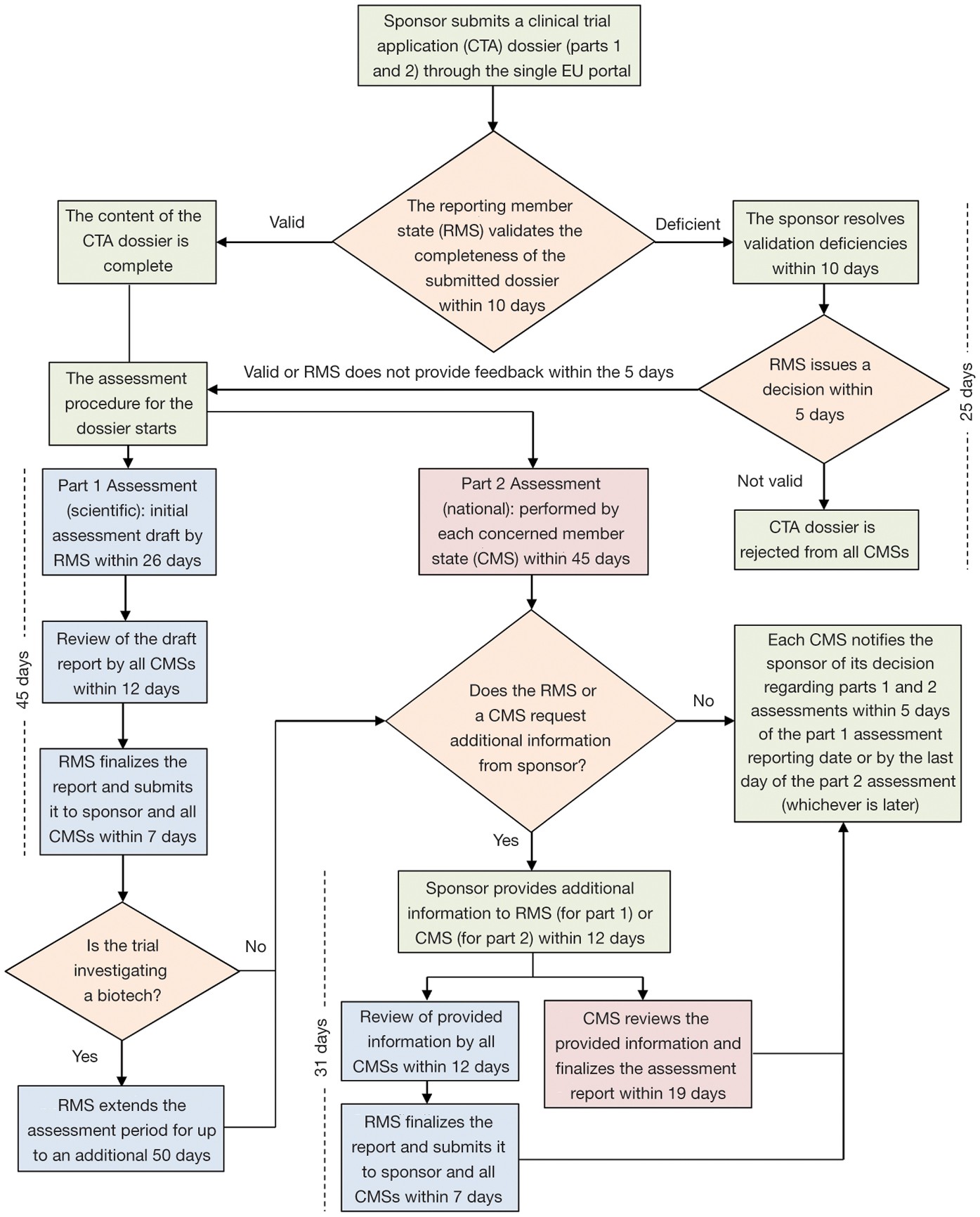
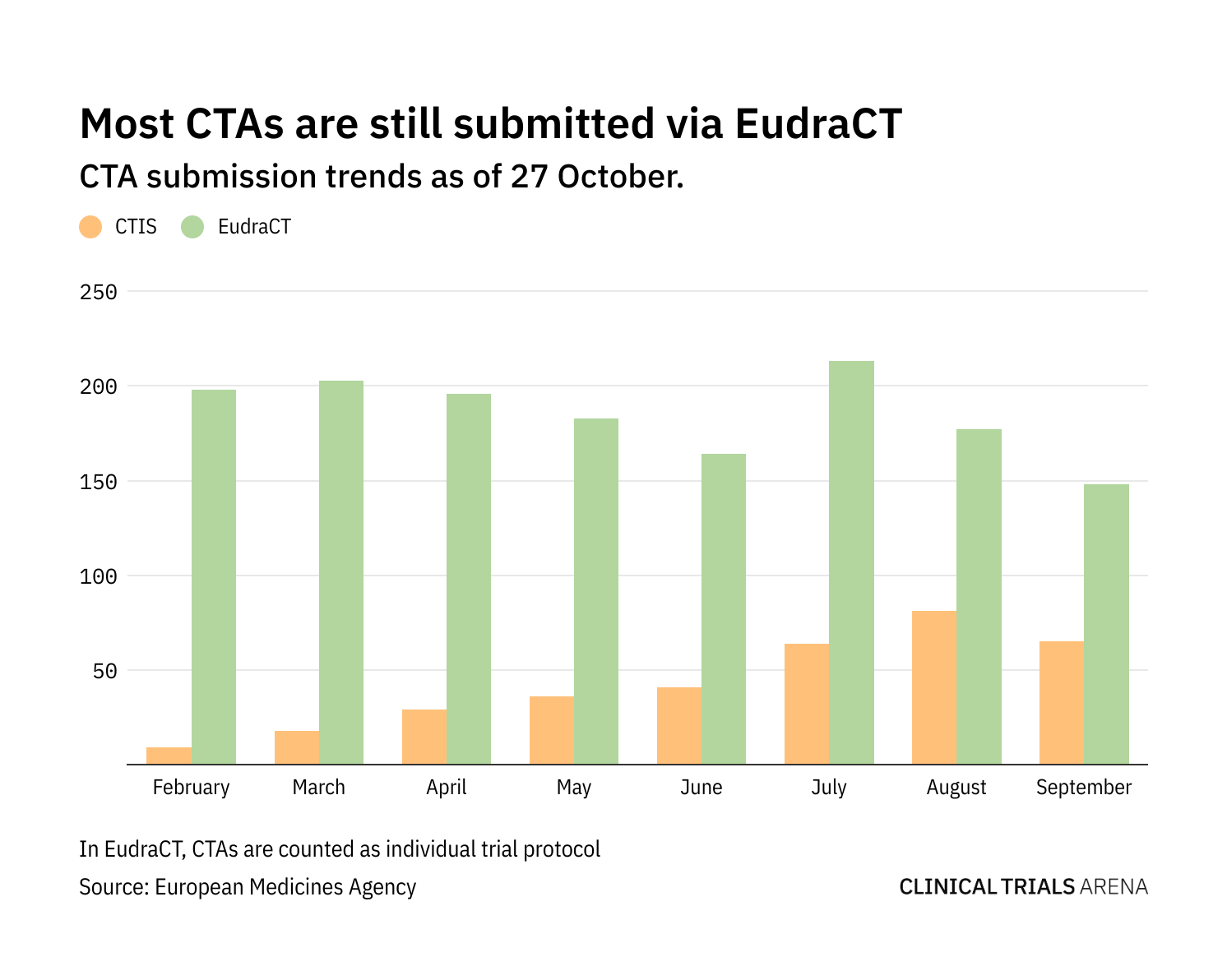
Regulatory Affairs 101: Introduction to Investigational New Drug Applications and Clinical Trial Applications - Chiodin - 2019 - Clinical and Translational Science - Wiley Online Library

Amazon.com: The Four Villains of Clinical Trial Agreement Delays and How to Defeat Them: Addressing CTA Delays Comprehensively: 9781973622697: Araujo, Débora S.: Books