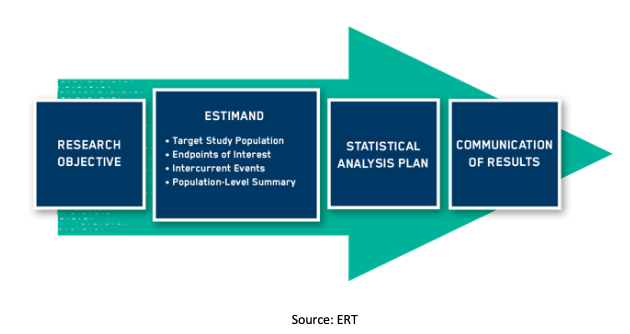
Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines: Brody PhD, Tom: 9780123919113: Amazon.com: Books

EU clinical research framework. ICH GCP = International Conference on... | Download Scientific Diagram

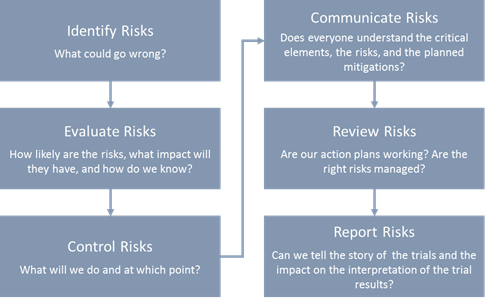















(135).jpg)