Idarucizumab for dabigatran reversal: the first 6 months in a tertiary centre - Wheeler - 2019 - Internal Medicine Journal - Wiley Online Library

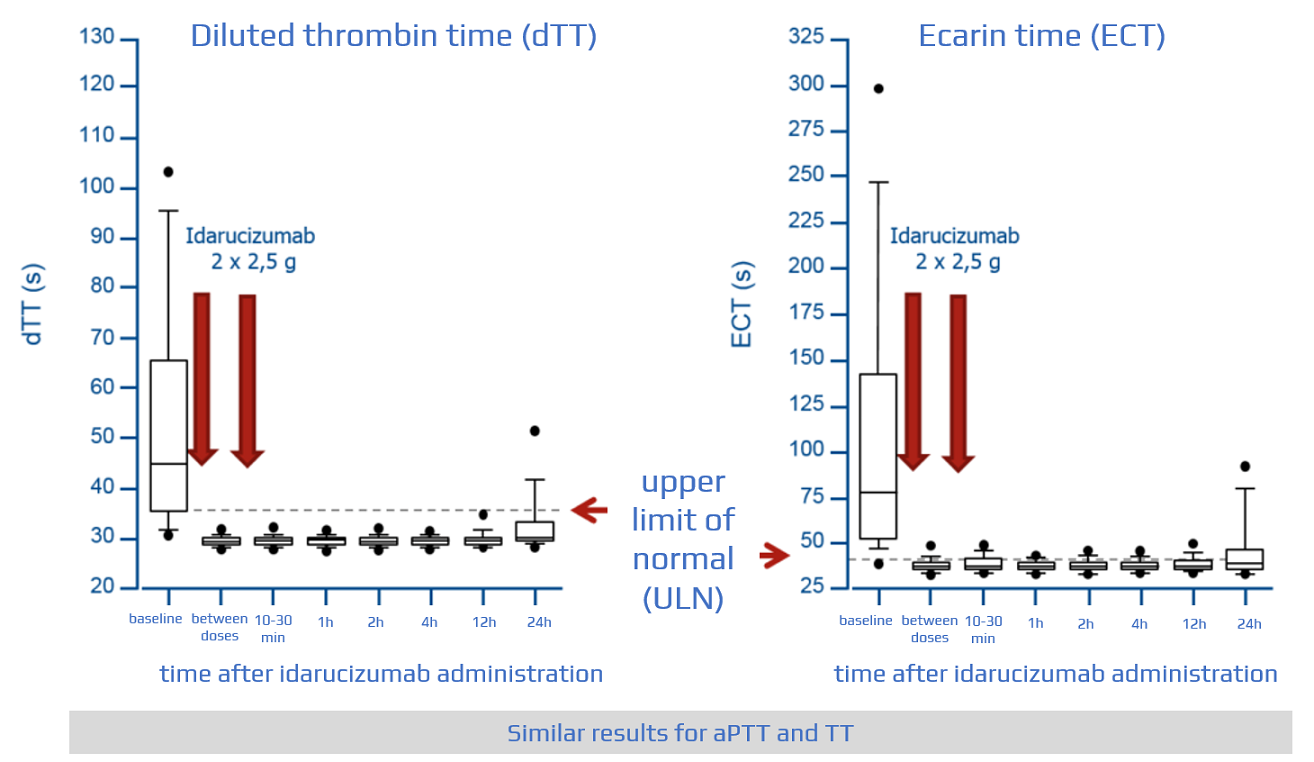
Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: a randomised, placebo-controlled, double-blind phase 1 trial - The Lancet
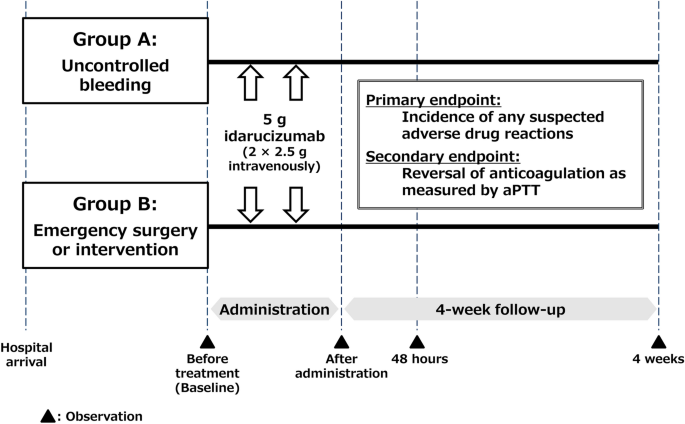
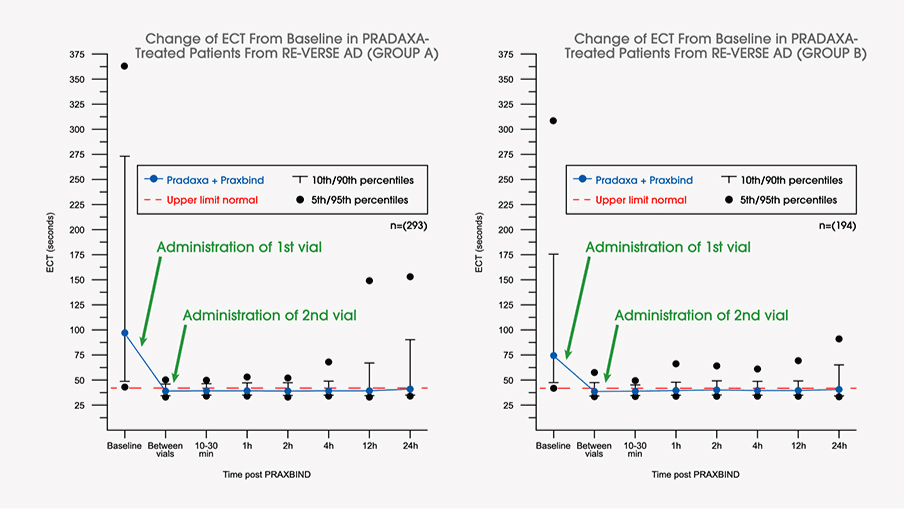
Idarucizumab for Emergency Reversal of Anticoagulant Effects of Dabigatran: Interim Results of a Japanese Post-Marketing Surveillance Study | SpringerLink
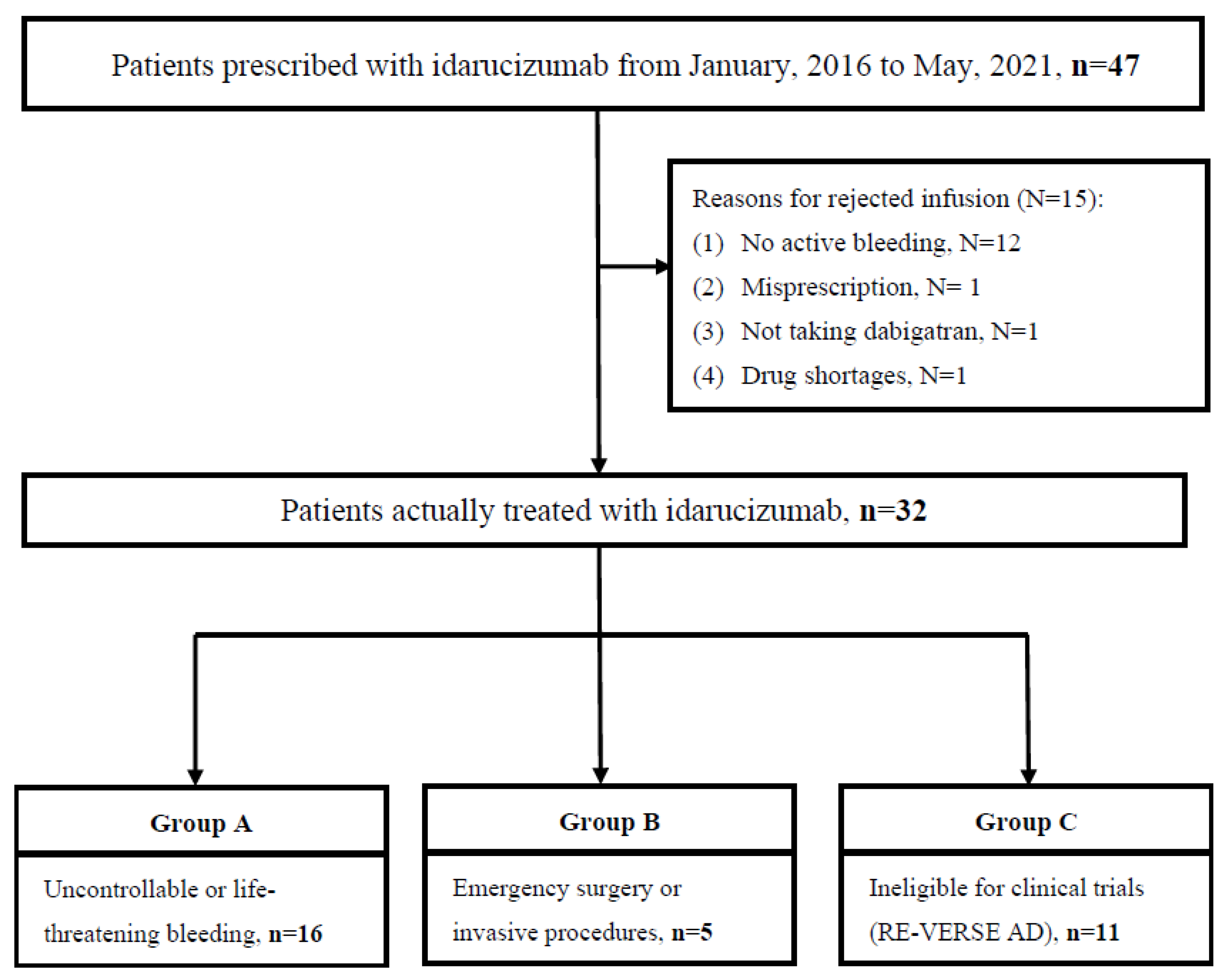
Medicina | Free Full-Text | Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials

PDF) Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials

Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: a randomised, placebo-controlled, double-blind phase 1 trial - The Lancet

The Renal Elimination Pathways of the Dabigatran Reversal Agent Idarucizumab and its Impact on Dabigatran Elimination - Stephan Glund, Guanfa Gan, Viktoria Moschetti, Paul Reilly, Markus Honickel, Oliver Grottke, Joanne Van Ryn,
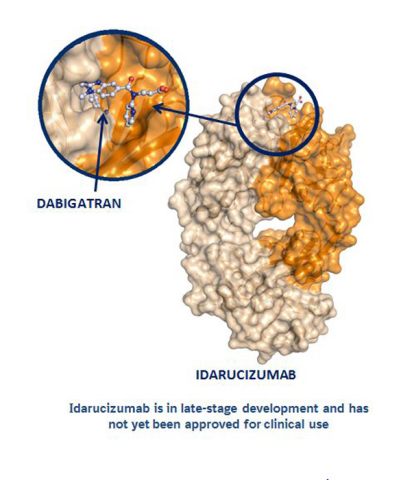
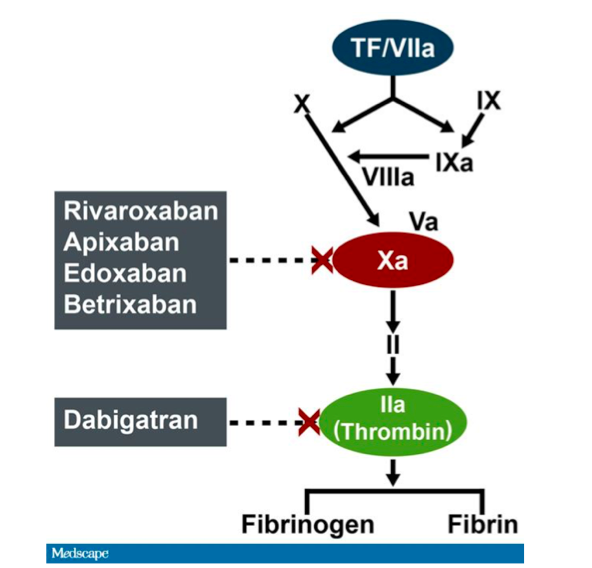
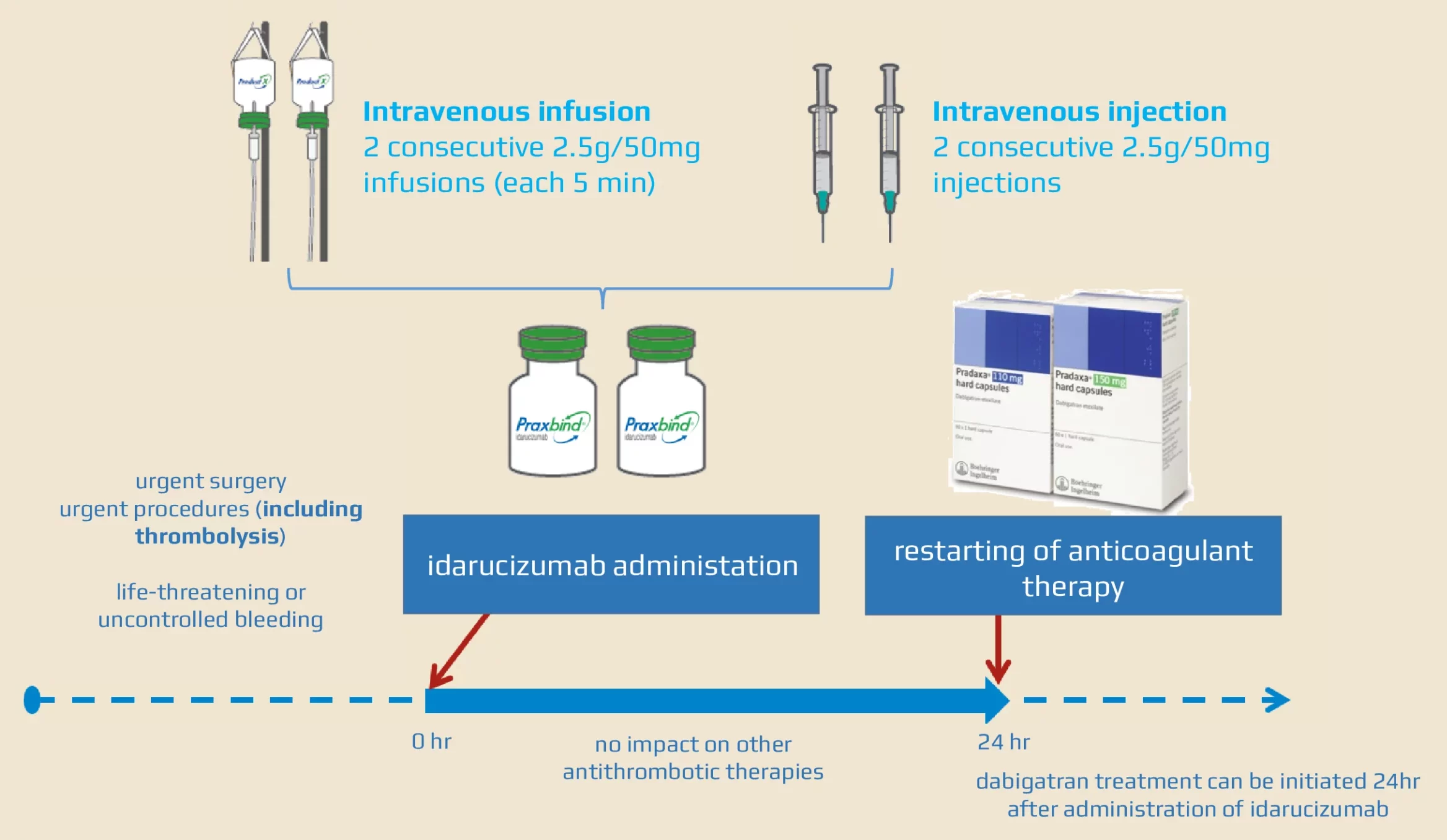
Idarucizumab, a Specific Reversal Agent for Dabigatran: Mode of Action, Pharmacokinetics and Pharmacodynamics, and Safety and Ef
Interpretation of idarucizumab clinical trial data based on spontaneous reports of dabigatran adverse effects in the French phar

Medicina | Free Full-Text | Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials

FDA Approves Praxbind® (idarucizumab), Specific Reversal Agent for Pradaxa® (dabigatran etexilate) | Business Wire

These highlights do not include all the information needed to use PRAXBIND safely and effectively. See full prescribing information for PRAXBIND. PRAXBIND® (idarucizumab) injection, for intravenous useInitial U.S. Approval: 2015
Usefulness of initial plasma dabigatran concentration to predict rebound after reversal | Haematologica

Design of the Reversal Effects of Idarucizumab on Active Dabigatran... | Download Scientific Diagram
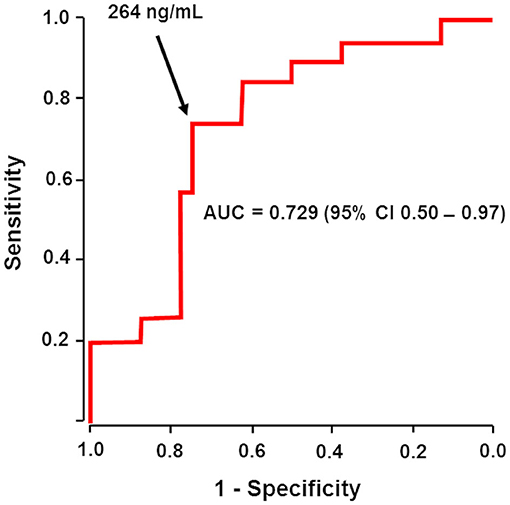
Frontiers | Dabigatran Level Before Reversal Can Predict Hemostatic Effectiveness of Idarucizumab in a Real-World Setting