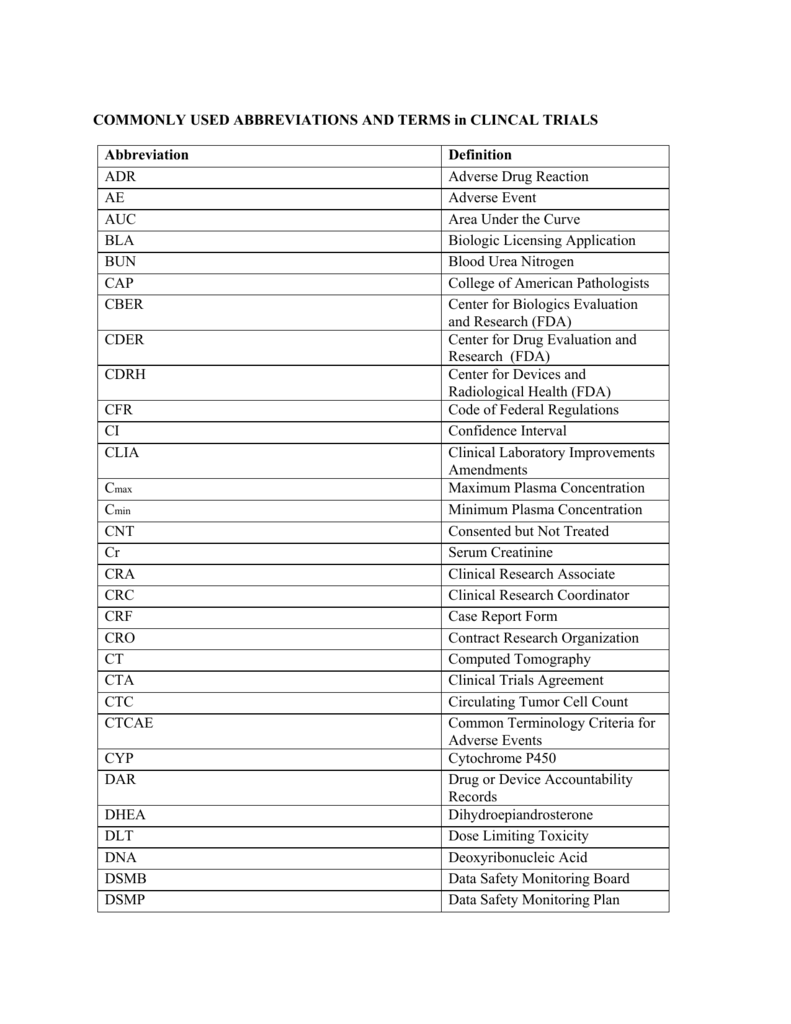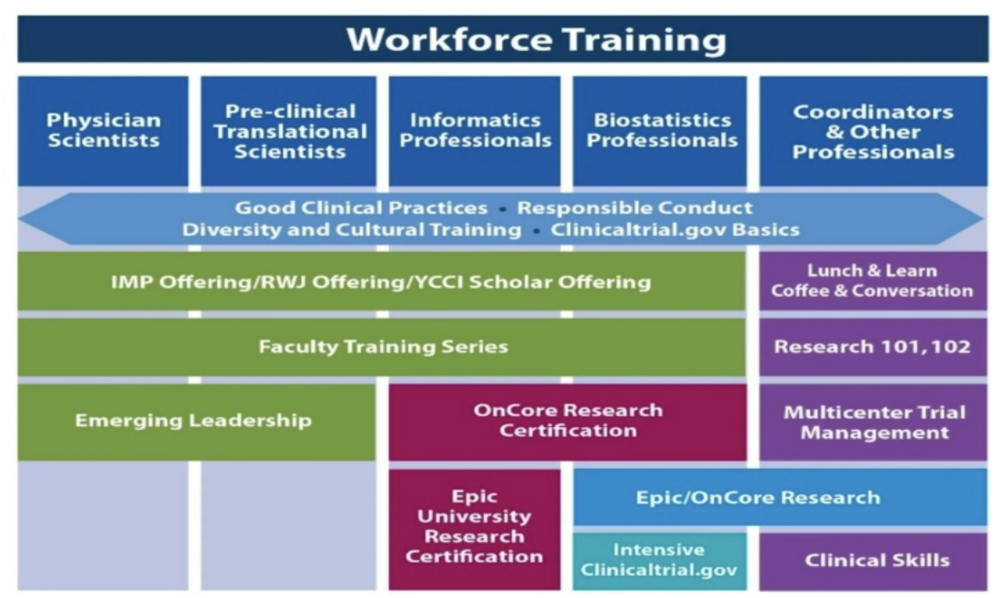
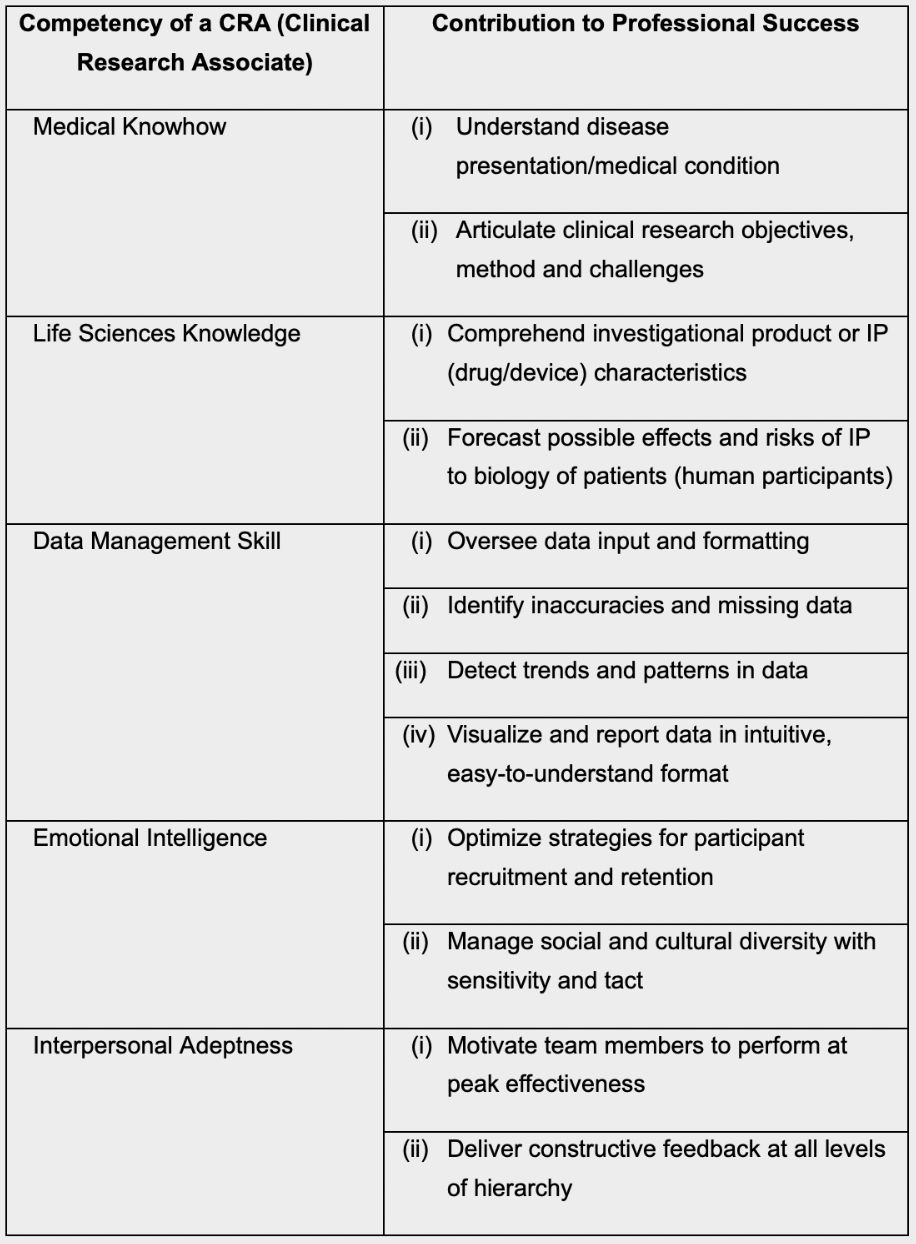
CRO — Clinical Research Blog | Certified Clinical Research Professionals Society - Clinical Research Certification

The neutrophil–lymphocyte ratio and its utilisation for the management of cancer patients in early clinical trials | British Journal of Cancer

fda guidance on conduct of clinical trials of medical products during covid-19 pandemic — Clinical Research Blog | Certified Clinical Research Professionals Society - Clinical Research Certification
fda guidance on conduct of clinical trials of medical products during covid-19 pandemic — Clinical Research Blog | Certified Clinical Research Professionals Society - Clinical Research Certification

Abbreviations-integumentary system in Medical Terminology(Adaptive*) Tutorial 22 May 2022 - Learn Abbreviations-integumentary system in Medical Terminology(Adaptive*) Tutorial (14970) | Wisdom Jobs India
Clinical Study Protocol with Amendment 04 A Multicenter, Multinational, Randomized, Double-Blind, Placebo-Controlled, Parallel-