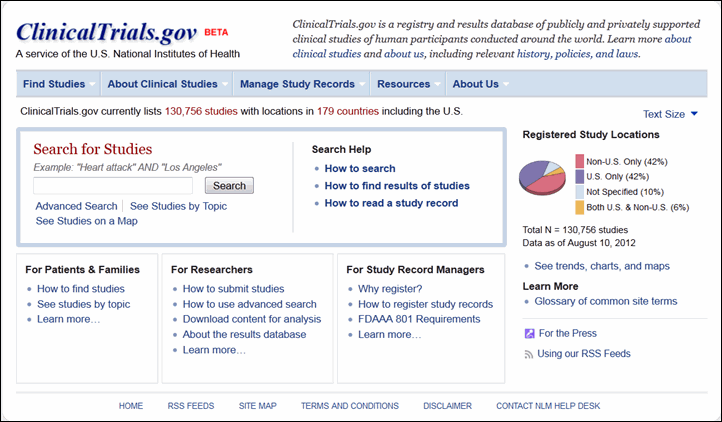
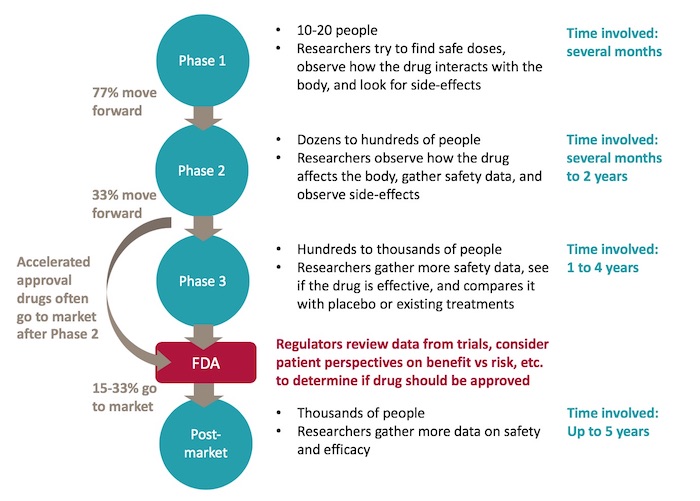
Many trials of hydroxychloroquine for SARS-CoV-2 were redundant and potentially unethical: an analysis of the NIH clinical trials registry - Journal of Clinical Epidemiology

Don't Jeopardize Future Funding or Publication: Register and Upload Your Clinical Trial Data Now - Dean's Office Blog

Key Compliance Risks in Clinical Trials Kathleen Meriwether Principal, ERNST & YOUNG, LLP Fraud Investigation & Dispute Services. - ppt download

PDF) Many Trials of Hydroxychloroquine for SARS-CoV-2 Were Redundant and Potentially Unethical: An Analysis of the NIH Clinical Trials Registry

The evolution in registration of clinical trials: a chronicle of the historical calls and current initiatives promoting transparency | Semantic Scholar

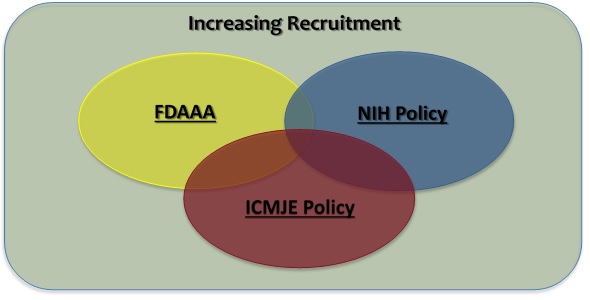
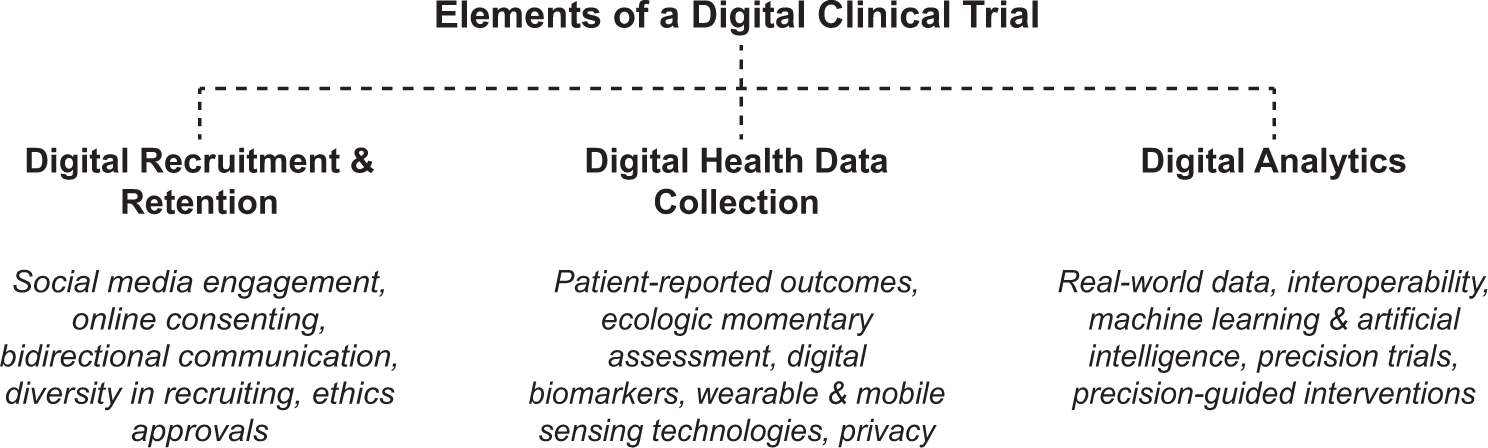
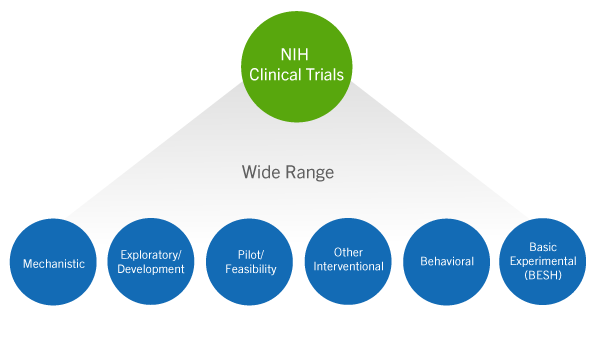
Requirements for Registering & Reporting NIH-funded Clinical Trials in ClinicalTrials.gov | grants.nih.gov